FOR IMMEDIATE RELEASE
FOR MORE INFORMATION CONTACT:
Dr. Stephen H. Wolpo, DDS
Smile Sensations
(203) 323-9277
steve@smilesensations.com
STAMFORD DENTIST OFFERS REVOLUTIONARY, “NEEDLE-FREE” PAINLESS ANESTHETIC ALTERNATIVE
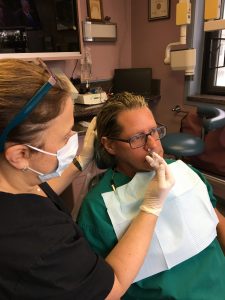 Stamford, CT, January 20, 2016…. Smile Sensations, the office of Dr. Stephen H. Wolpo, DDS, will begin offering its patients a new, FDA -approved topical dental anesthetic that is administered through a nasal spray, replacing traditional needle injections for many restorative procedures.
Stamford, CT, January 20, 2016…. Smile Sensations, the office of Dr. Stephen H. Wolpo, DDS, will begin offering its patients a new, FDA -approved topical dental anesthetic that is administered through a nasal spray, replacing traditional needle injections for many restorative procedures.
“One of the most stressful experiences for adults and children alike is fear of the needle and the numbness of the lips that can follow for several hours,” said Dr. Wolpo. “In some cases, their anxiety over receiving a shot of novocaine is so overwhelming and debilitating that it leads them to postpone dental treatments that are vital for maintaining their overall health. Intranasal drugs are already being used successfully in other medical specialties, and I am so excited to now be able to introduce this ‘no shot’ technology to my patients. At Smile Sensations, we remain committed to offering the latest advancements in dental care to help create a more calming, comfortable and positive dental experience.”
Smile Sensations, located at 60 Strawberry Hill Avenue in Stamford, was also one of the first dental offices in Fairfield County to offer “drill-free” painless laser dentistry and non-invasive velscope screening for oral cancer.
Kovanaze Nasal Spray (tetracaine HCL and oxymetazoline HCL), developed by St. Renatus LLC, is the world’s first known local dental anesthetic administered through the nasal cavity, ending a century-old method of delivering dental anesthesia using a needle injection. It is indicated for use in adults age 18 and older and children who weigh approximately 90 pounds or more and for procedures involving most of the upper teeth. It received U.S. Food and Drug Administration approval on June 29, 2016.
 (203) 399-0707
(203) 399-0707 Our Location
Our Location